Contents
[1. Introduction] [2. Results and Discussion]
[3. Conclusions] [Bibliographic references]
Department of Geology, University of Tartu, Ravila 14A, EE-50411 Tartu (Estonia)
Faculty of Earth Sciences, University of Silesia, Będzińska 60, PL-41-200 Sosnowiec (Poland)
Manuscript online since March 12, 2012
[Editor: Christian ]
Cloudina, an important Ediacaran index fossil, is considered as one of the earliest biomineralizing organisms. Its biological affinities have not been fully resolved and phylogenetic links with both annelids and cnidarians have traditionally been suggested. Differences in tube morphology, ultrastructure and biomineralization suggest that Cloudina is not closely related to any recent skeletal annelid (e.g., serpulids, sabellids and cirratulids) and their skeletons are not homologous. The way of asexual reproduction in Cloudina resembles more that of cnidarians. The presence of a closed tube origin (base) in Cloudina is also compatible with the hypothesis of an animal of cnidarian grade.
Cloudina; Annelida; Serpulidae; Cnidaria; Ediacaran; biomineralization.
O. & M. (2012).- Inconsistencies in proposed annelid affinities of early biomineralized organism Cloudina (Ediacaran): structural and ontogenetic evidences.- Carnets de Géologie [Notebooks on Geology], Brest, Article 2012/03 (CG2012_A03), p. 39-47.
Incohérences dans les affinités supposées de l'organisme primitif biominéralisé Cloudina (Ediacaran) avec les annélides : preuves relevant de la structure et du développement ontogénétique.- Cloudina, un important fossile édiacarien, est considéré comme l'un des premiers organismes biomineralisés. Ses affinités biologiques n'ont pas encore été entièrement élucidées : des relations phylogénétiques avec les annélides et les cnidaires sont habituellement envisagées. Des différences dans la morphologie, l'ultrastructure et la biominéralisation du tube suggèrent que Cloudina ne puisse être étroitement apparentée à aucun squelette d'annélides actuelles, qu'il s'agisse de serpulidés, de sabellidés ou de cirratulidés ; leurs squelettes ne sont pas homologues. Le mode de reproduction asexuée de Cloudina ressemble davantage à celui des cnidaires. De plus, la configuration fermée de la partie initiale (proximale) du tube chez Cloudina est compatible avec l'hypothèse d'un animal de la catégorie des cnidaires.
Cloudina ; Annelida ; Serpulidae ; Cnidaria ; Édiacarien ; biominéralisation.
Cloudina is a tube-building metazoan organism of problematic affinities that widely occurs in deposits of the terminal Neoproterozoic Ediacaran Period (, 1990), between 549 ± 1 Ma and 542 ± 1 Ma ( et al., 1995; et al., 2003). Two distinct species are recognized, C. hartmannae , 1972, and C. carinata et al., 2010, while rest of species could be junior synonyms of C. hartmannae ( et al., 2010). It is significant for being among the precursors of biologically controlled mineralization and being an important Ediacaran index fossil ( et al., 2005).
The geochemical data suggest that Cloudina formed originally aragonitic skeleton ( & , 2005), however, relatively little is known about the biology of Cloudina. There is a record of drilling predation upon Cloudina ( & , 1992; et al., 2003). Followed from the location of predatory drill holes, Cloudina organism was presumably able to protect its shell nearest to the aperture and its soft body may have been located in the upper, growing portion of the tube ( et al., 2003).
According to traditional point of view, Cloudina was closely related to annelids and especially to serpulids (, 1972; , 1976). However, & (1985) and et al. (1990) did not support 's and 's proposed relationships, and classified Cloudina just in its own family, Cloudinidae. The occurrence of dichotomous branching in C. hartmannae, which implies asexual reproduction led (1990) to classify Cloudina as a coral-like cnidarian. Recently, dichotomous branching has also been found in C. carinata et al., 2010, from the Ediacaran of Spain ( et al., 2010). In contrary, as a result of their thorough review on biology of Cloudina, et al. (2005) found that Cloudina's morphology, skeletogenesis, and asexual reproduction are broadly similar to modern serpulid annelids, indicating possible phylogenetic relationships or morphological convergence. They also concluded that traditional hypothesis of Cloudina's annelid affinities remains plausible.
The earliest definitive annelids with biomineral skeletons (calcitic) were machaeridians in the Early Ordovician, which had a body covered with the calcareous plates ( et al., 2008), but they did not build calcareous tubes. The earliest hitherto known calcareous annelid tubeworms are serpulids (Serpulidae) and their definitive representatives are known from the Triassic ( et al., 2008c, 2008a; & , 2009). The calcareous tubes also occur in sabellid (Sabellidae) and cirratulid (Cirratulidae) polychaetes, since the Early Jurassic (, 2004; et al., 2008a) and the Oligocene respectively ( et al., 2000; , 2009). Recent discoveries on the skeletal structures and biomineralization of calcareous polychaetes (, 1994; , 2005, 2007, 2008, 2009; et al., 2008c, 2008a, 2008b, 2008d, 2009; , 2009; & , 2009; et al., 2010) allows us for the first time to make a complete comparison with the skeletal structures and skeletogenesis of problematic tubicolous fossils such as Cloudina.
The aims of this paper are:
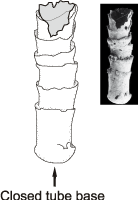
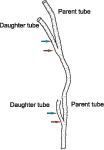
Cloudina hartmannae tube has a closed origin ( et al.,
2003, 2005: fig. 1B): Fig. 1 ![]() . C. hartmannae ( et al.,
2003, 2005: fig. 1A-B) and C. carinata ( et al.,
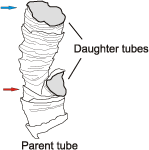
2010: figs. 4-6) tubes do not have flattened attachment surfaces. The diameter of C. hartmannae shell could increase relatively rapidly after dichotomous branching ( et al.,
2003: fig. 3E, 2005: fig. 1P): Fig. 2
. C. hartmannae ( et al.,
2003, 2005: fig. 1A-B) and C. carinata ( et al.,
2010: figs. 4-6) tubes do not have flattened attachment surfaces. The diameter of C. hartmannae shell could increase relatively rapidly after dichotomous branching ( et al.,
2003: fig. 3E, 2005: fig. 1P): Fig. 2 ![]() . The tube diameter of both daughter tubes could be about equal and smaller than the diameter of parent tube
( et al., 2005: fig.
1K-L, 2007: fig. 6.5) in C. hartmannae (Figs. 2
. The tube diameter of both daughter tubes could be about equal and smaller than the diameter of parent tube
( et al., 2005: fig.
1K-L, 2007: fig. 6.5) in C. hartmannae (Figs. 2 ![]() - 3
- 3 ![]() )
and different in C. carinata ( et al.,
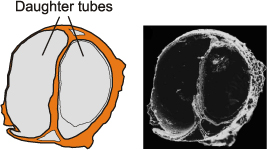
2010: fig. 8). The shape of C. hartmannae tube cross section changes during dichotomous branching from circular (parent tube) to semi-circular,
both daughter tubes ( et al.,
2005: fig. 1L): Fig. 3
)
and different in C. carinata ( et al.,
2010: fig. 8). The shape of C. hartmannae tube cross section changes during dichotomous branching from circular (parent tube) to semi-circular,
both daughter tubes ( et al.,
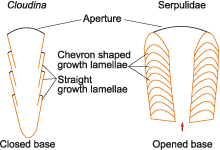
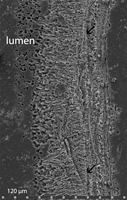
2005: fig. 1L): Fig. 3 ![]() . Growth lamellae of C. hartmannae tube are long and straight in longitudinal section
- it lacks chevron shaped growth lamellae - ( et al.,
2005: fig. 1C): Fig. 4
. Growth lamellae of C. hartmannae tube are long and straight in longitudinal section
- it lacks chevron shaped growth lamellae - ( et al.,
2005: fig. 1C): Fig. 4 ![]() . Tube wall has a microgranular homogeneous ultrastructure ( et al.,
2003; et al., 2003: fig.
3O, 2005: fig. 1H): Fig. 5a
. Tube wall has a microgranular homogeneous ultrastructure ( et al.,
2003; et al., 2003: fig.
3O, 2005: fig. 1H): Fig. 5a ![]() .
.
Click on thumbnail to enlarge the image.
Figure 1: Cloudina hartmannae from Dengying Formation, closed bulbous basal end (SEM photo reprinted by permission of publisher from et al. (2005: fig. 1A).
Click on thumbnail to enlarge the image.
Figure 2: Cloudina hartmannae from Dengying Formation, dichotomous branching, showing rapid increase of daughter tubes diameter (arrows), drawn after et al. (2003: fig. 3E).
Click on thumbnail to enlarge the image.
Figure 3: Cross section of daughter tubes within the parent tube, note the semicircular cross section of daughter tubes (SEM photo reprinted by permission of publisher from et al. (2005: fig. 1L).
a. Discussion of characters
Cloudina has a tubicolous shell that somewhat resembles tubes of calcareous polychaete annelids
(e.g., serpulids, sabellids and cirratulids) in its probably calcareous original composition, smooth tube lumen and presence of peristome-like structures on the shells external surface (Figs. 1
![]() - 2
- 2 ![]() ). Cloudina (Fig. 2
). Cloudina (Fig. 2 ![]() ) also resembles some serpulids (Fig. 6
) also resembles some serpulids (Fig. 6 ![]() ) and calcareous Cirratulidae ( et al.,
2000) by its occurrence of asexual reproduction (,
2001; ten & ,
2009). et al.
(2005) found that there are following differences between Cloudina and serpulids: a) serpulid tube walls are thicker and solid, and they do not consist of nested funnels; b) the dichotomous branches of serpulids are not as deeply nested within the parent tube as in Cloudina. In addition, we found several other important differences between the calcareous polychaete tubes and Cloudina:
) and calcareous Cirratulidae ( et al.,
2000) by its occurrence of asexual reproduction (,
2001; ten & ,
2009). et al.
(2005) found that there are following differences between Cloudina and serpulids: a) serpulid tube walls are thicker and solid, and they do not consist of nested funnels; b) the dichotomous branches of serpulids are not as deeply nested within the parent tube as in Cloudina. In addition, we found several other important differences between the calcareous polychaete tubes and Cloudina:
1) Ultrastructure - Annelids do not have microgranular tube ultrastructure. Their primitive tube ultrastructure is either spherulitic
- sabellids - ( et al.,
2008a), spherulitic prismatic - sabellids and
cirratulids - ( et al.,
2008a; , 2009) or composed of irregularly oriented elongate prisms
- serpulids - ( et al., 2008c,
2008b). The presence of microgranular tube structure in serpulids is an artifact of sample natural
breakage surface. Unpolished and not etched samples of serpulids with irregularly oriented prismatic structure seem to be composed of homogeneous fine granules under SEM (O. ,
pers. obs.). The tube ultrastructure of asexually reproducing Salmacina and Filograna, compared to Cloudina by et al.
(2005), is not fine microgranular, but composed of irregularly oriented elongate prisms in a homogeneous matrix (see et al.,
2008b): Fig. 5b ![]() . However, ultrastructure of organic-rich tabulae (= transverse cross walls) inside the Serpula vermicularis tube is indeed similar to that of Cloudina, but they are formed in a different process than the tube wall ( et al.,
2008b). However, the original structure of Cloudina may have not been microgranular, as different phosphatized calcareous skeletons and organic substrates could show this kind of fabric.
. However, ultrastructure of organic-rich tabulae (= transverse cross walls) inside the Serpula vermicularis tube is indeed similar to that of Cloudina, but they are formed in a different process than the tube wall ( et al.,
2008b). However, the original structure of Cloudina may have not been microgranular, as different phosphatized calcareous skeletons and organic substrates could show this kind of fabric.
Click on thumbnail to enlarge the image.
Figure 4: Schematic line drawings of Cloudina and serpulid tubes, showing morphology of growth lamellae and tube base.

Click on thumbnail to enlarge the image.
Figure 5: a.- Microgranular ultrastructure of Cloudina sinensis, Gaojiashan Member of Dengying formation, Ningqiang, Shaanxi, China (after et al., 2003: fig. 1b). b.- Irregularly oriented prismatic structure in cross section in Salmacina incrustans, ZMA V.Pol.3814, Costa Brava, Spain.
2) Biomineralization - et al.
(2005) found that Cloudina tubes do not show radial fibrous microstructure or palisade orientation of crystals, crystal nucleation and growth on a sheeted organic or inorganic substrate did not play a significant role in Cloudina skeletogenesis. They concluded that crystals were probably precipitated by skeleton-secreting organs and mixed with an organic matrix, what is similar to serpulids (,
1958; , 1971; & ,
1975). However, standard granular secretion model of serpulids (,
1958; , 1971) has recently been cast in doubt by the discovery of complex oriented tube structure in Pomatoceros americanus ( et al.,
2009) on which the model of serpulid secretion was based. Several other discoveries on the skeletal ultrastructures in serpulids contradict the traditional point of view on their biomineralization ( et al.,
2008a, 2008b, 2008d). New evidence on the skeletal structures of serpulids suggests that serpulids were organic matrix-mediated biomineralizers similar to that of molluscs ( et al.,
2008a, 2008b, 2008d,
2009; et al.,
2010). The biomineralization of sabellids (Fig. 7 ![]() ) and cirratulids produces oriented prismatic structures, perpendicular to organic sheets, which cannot be achieved in a granular secretion process, as well ( et al.,
2008a; , 2009).
) and cirratulids produces oriented prismatic structures, perpendicular to organic sheets, which cannot be achieved in a granular secretion process, as well ( et al.,
2008a; , 2009).
Click on thumbnail to enlarge the image.
Figure 6: Salmacina amphidentata (Serpulidae), Indian River Lagoon in the vicinity of Fort Pierce Inlet, Florida. Detail of an aggregation showing periodic branching of the tubes. Note that diameter of the daughter tubes increases very slowly after branching (arrows). Drawn after (2001: p. 110, fig. 1B).
Click on thumbnail to enlarge the image.
Figure 7: Glomerula piloseta (Sabellidae), Recent, Lizard Island, Australia, polished and etched longitudinal section, showing aragonitic spherulitic prismatic structure. Arrows point to long subparallel to tube walls growth lamellae.
3) Morphology of growth lamellae - Growth lamellae are much longer than in serpulids (,
1994; , 1996; & ,
2008) in the longitudinal section, resembling those of tubicolous molluscs such as vermetid gastropods or scaphopods. Unlike the straight growth lamellae of Cloudina, most of serpulids have chevron shaped growth lamellae (,
1994; & ,
2008): Fig. 8 ![]() . Thus, tube formation in Cloudina should have been different from that of anterior accretion of serpulid tubes. However, growth lamellae of sabellids
- Glomerula - (Fig. 7
. Thus, tube formation in Cloudina should have been different from that of anterior accretion of serpulid tubes. However, growth lamellae of sabellids
- Glomerula - (Fig. 7 ![]() ) and cirratulids can be much longer in longitudinal section than in serpulids and thus resemble more those of Cloudina. The growth lamellae of Yangtzitubus (Ediacaran) are slightly similar to those of Cloudina ( et al.,
2009).
) and cirratulids can be much longer in longitudinal section than in serpulids and thus resemble more those of Cloudina. The growth lamellae of Yangtzitubus (Ediacaran) are slightly similar to those of Cloudina ( et al.,
2009).

Click on thumbnail to enlarge the image.
Figure 8: Pomatoceros triqueter (Serpulidae), Recent, Tjärnö, Sweden, polished section treated with solution, showing chevron shaped growth lamellae.
4) Morphology of tube origin - Embryonic serpulid tubes are opened at both ends (ten & van den ,
1993; & ,
2006): Figs. 4 ![]() & 9
& 9 ![]() , in contrast to closed tube origin of C. hartmannae ( et al.,
2003,
2005): Figs. 1
, in contrast to closed tube origin of C. hartmannae ( et al.,
2003,
2005): Figs. 1 ![]() & 4
& 4 ![]() . In C. carinata tube origin is reported to be open, but this was based on a fragmentary material, which may not represent complete tubes ( et al.,
2010). Serpulid tubes are often closed off by the tabulae (see ,
2009, and references therein). Similarly, tubes of Glomerula (Sabellidae) have presumably an open origin because of similar tube formation. It is likely that also calcareous cirratulid tubes are not closed at their origin. Thus, the beginning of tube formation in Cloudina should fundamentally differ from that of serpulids and probably also from the other calcareous polychaetes.
. In C. carinata tube origin is reported to be open, but this was based on a fragmentary material, which may not represent complete tubes ( et al.,
2010). Serpulid tubes are often closed off by the tabulae (see ,
2009, and references therein). Similarly, tubes of Glomerula (Sabellidae) have presumably an open origin because of similar tube formation. It is likely that also calcareous cirratulid tubes are not closed at their origin. Thus, the beginning of tube formation in Cloudina should fundamentally differ from that of serpulids and probably also from the other calcareous polychaetes.

Click on thumbnail to enlarge the image.
Figure 9: Serpulid Spirorbis spirorbis, substrate cemented side, showing an open tube apex (arrow), Recent, North Sea.
5) Shape of tube cross section - Semicircular cross section of daughter tubes lumen inside the parent tube in Cloudina (Fig. 3 ![]() ) differs from that of circular lumen cross section of serpulid and sabellid tubes. Tubes with semicircular cross section occur in Yangtzitubus semiteres et al.
(2009) from the Ediacaran (Doushantuo Formation) of southern China ( et al.,
2009), but differently from Cloudina, their tubes are never circular in cross section. A semicircular cross section is also known in Conotubus & in
(1986) from the Dengying Formation (Ediacaran) of southern China ( et al.,
2007; et al.,
2009).
) differs from that of circular lumen cross section of serpulid and sabellid tubes. Tubes with semicircular cross section occur in Yangtzitubus semiteres et al.
(2009) from the Ediacaran (Doushantuo Formation) of southern China ( et al.,
2009), but differently from Cloudina, their tubes are never circular in cross section. A semicircular cross section is also known in Conotubus & in
(1986) from the Dengying Formation (Ediacaran) of southern China ( et al.,
2007; et al.,
2009).
6) Expansion of the tube - Rapid increase of tube diameter after dichotomous branching in Cloudina (Fig. 2 ![]() ) is different from that of slow increase in serpulids (see ,
2001: figs. 1B & 4A): Fig. 6
) is different from that of slow increase in serpulids (see ,
2001: figs. 1B & 4A): Fig. 6 ![]() . Cloudina could have a tube divergence angle about 30° that is not possible in case of serpulid or any other calcareous polychaete tubes (personal obs. O. ). Such a rapid increase in diameter of tube, probably reflecting the increase of soft body volume would have prevented the deep retraction of soft body into the tube possibly in all calcareous polychaete tubeworms. Rapid increase of tube diameter after dichotomous branching in Cloudina somewhat resembles Ramitubus increscens ( et al.,
2008: pl. 1, fig. 6) interpreted as cnidarian ( et al.,
2002; et al., 2008) or alternatively
as algae (, 2005).
. Cloudina could have a tube divergence angle about 30° that is not possible in case of serpulid or any other calcareous polychaete tubes (personal obs. O. ). Such a rapid increase in diameter of tube, probably reflecting the increase of soft body volume would have prevented the deep retraction of soft body into the tube possibly in all calcareous polychaete tubeworms. Rapid increase of tube diameter after dichotomous branching in Cloudina somewhat resembles Ramitubus increscens ( et al.,
2008: pl. 1, fig. 6) interpreted as cnidarian ( et al.,
2002; et al., 2008) or alternatively
as algae (, 2005).
7) Change of tube diameter - Diameter of both daughter tubes is smaller than that of parent tube (before branching) after the dichotomous branching in Cloudina ( et al., 2005: fig. 1K-L, 2007: fig. 6.5). It is different from that of serpulids where parent tube diameter does not decrease after budding (, 2001: fig. 4A). Similarly to Cloudina tube diameter of both daughter tubes is smaller than the parent one in Ramitubus increscens ( et al., 2008: pl. 1, fig. 6).
8) Attachment surfaces - A lack of flattened attachment surfaces in Cloudina tube differs from that of tubes of calcareous polychaetes. Almost all known calcareous polychaetes begin their growth cemented to the substrate with the exception of serpulid Ditrupa (ten & , 1990). However, the tube of Ditrupa is opened from both ends and it has chevron shaped growth lamellae, two layered tube with outer layer having an oriented ultrastructure ( et al., 2008d). Phylogenetically, Ditrupa is among the most derived serpulids and does not represent the plesiomorphic condition for serpulids (ten , 2008, personal comm.). The plesiomorphic serpulids were all substrate cemented at least in the beginning of their growth.
b. Affinities of Cloudina
Only tubicolous shell, smooth lumen and presence of peristomes in Cloudina are unequivocally similar to that of serpulids (Table 1). et al. (2011) found some similarity between sabellid tubes constructed of thin organic films and aragonitic layers. However, there are strong differences between Cloudina and other calcareous polychaetes (e.g., sabellids and cirratulids). Cloudina differs from serpulids in the tube ultrastructure, proposed type of biomineralization, closed tube origin (base), semicircular cross section of daughter tubes, rapid increase of the tube diameter after dichotomous branching, change of tube diameter, and lack of attachment surfaces. These differences in tube morphology, ultrastructure and biomineralization indicate that Cloudina is not closely related to any recent skeletal annelid (e.g., serpulids, sabellids and cirratulids) and their skeletons are not homologous (Table 1). However, taking into account that annelids are a very diverse group of invertebrates and biomineralization can appear and disappear several times within the phyla (, 2008) it is impossible to entirely rule out the annelids as potential close relatives to Cloudina.
Characters |
Cloudina | Calcareous polychaetes (Serpulidae, Sabellidae, Cirratulidae) |
Cnidaria |
| Tubicolous shell | x | x | x |
| Smooth lumen | x | x | x |
| Peristomes | x | x | x |
| Asexual reproduction | x | x | x |
| Granular microstructure | x | - | x |
| Closed tube base | x | - | x |
| Semicircular tube cross section | x | - | x |
| Rapid increase of the tube diameter | x | - | x |
| Diameter of daughter tubes about equal | x | - | x |
| Attachment surfaces lacking | x | - | x |
Table 1: Comparison of Cloudina characters with calcareous polychaetes and Cnidaria [ x : character present; - : character absent]
The following characters are easiest to understand if one assumes a cnidarian affinity for Cloudina (Table 1):
Thus, the way of asexual reproduction in Cloudina resembles closely that of cnidarians, especially anthozoans. et al. (2011) found that pattern of asexual reproduction of Cloudina that has been observed by et al. (2005: fig. 1K-R) matches well to intercalar budding of cnidarians but not to serpulid budding with a bud tube development at the surface of the parental one but not inside it (, 2001). The closed tube origin of Cloudina is also compatible with the hypothesis of an animal of cnidarian grade. There is a coral with conical shell (Cothonion) known from the Early Cambrian of Australia and North Greenland (, 2011). It shares some characters with the Cloudina, such as conical shape, closed base and fast increase of the diameter.
Alternatively, et al. (2011) found that the presence of both 'hemispherical basal end' and tabulae could favour a microconchid lophophorate affiliation for Cloudina. However, microconchids are evolutionarily derived encrusting tentaculitoid tubeworms, that appeared first in the Late Ordovician (, 2010), and so not the best candidates among tentaculitoids for comparison with Cloudina. The earliest known tentaculitoids were cornulitids, that presumably appeared in the Middle Ordovician ( & , 2009). If tentaculitoid tubeworms are indeed the stem group phoronids as proposed by et al. (2010), they may well have been present at least as early as in the Early Cambrian. However, early tentaculitoids probably had a plesiomorphic substrate cemented life mode ( & , 2009) that differs from life mode of unattached Cloudina. In addition, skeletal ultrastructures of tentaculitoids are much more advanced than that of Cloudina.
In contrast, the simple ultrastructure in Cloudina (et al., 2003; et al., 2005) does not give many clues for comparison with any particular invertebrate phylum as they all could have had such a plesiomorphic condition in the Ediacaran. In addition to Cloudina, tubes could branch in a few other Ediacaran tubicolous fossils ( et al., 2009). These are Ramitubus, Yangtzitubus and Waltheria et , 1999. Yangtzitubus has also slightly similar wall structure, and it lacks cross walls ( et al., 2009). Thus, among the non-biomineralizing tubicolous fossils of Ediacaran, Yangtzitubus could phylogenetically be most closely linked to Cloudina.
c. Paleobiology of Cloudina
et al. (2003) found that predatory borings in Cloudina are located at relatively uniform distance from the shell apertures that points to either control by the life orientation of the shells, such as the position of the sediment surface, or, more likely, an avoidance response by the predator to protective measures located near the aperture.They hypothesized that absence of boreholes further from the aperture implicates one or more of the following:
Cloudina is envisioned here as an animal that is not capable for much moving back and forwards in its tube, or not moving at all, because of the fast increase of tube diameter after the dichotomous branching. The Cloudina animal was probably also entirely attached to the tube wall with its soft body surface and it changed shape of its soft body cross section from circular before the asexual reproduction to semicircular after the asexual reproduction. Presumably, Cloudina was not retracting into the tube in asexual reproduction, but it was divided by lateral fission into two daughter animals which thereafter laterally secreted new tube walls around their soft body inside the parent tube (e.g., almost equal split of a cylindrical funnel into two half-cylinders in C. hartmannae).
We are grateful to the Paleontological Society for a Grant, to Estonian Science Foundation for grant ETF9064 and to the Estonian Ministry of Education and Science for the target project SF0180051s08. We thank Brian and an anonymous reviewer for constructive reviews.
J.E., J.P., S., S.A., J., M.W. & A. (2003).- Extinction of Cloudina and Namacalathus at the Precambrian-Cambrian boundary in Oman.- Geology, Boulder, vol. 31, p. 431-434.
S. (2005).- Mineralized skeletons and early animal evolution. In: D.E.G. (ed.), Evolving form and function: fossils and development.- Yale Peabody Museum Publications, New Haven, CT, p. 101-124.
S. & Z. (1992).- Predatorial borings in late Precambrian mineralized exoskeletons.- Science, Washington, vol. 257, p. 367-369.
R. & M. (2005).- A closer examination of biomineralization in the late Precambrian.- The Palaeontological Association 49th Annual Meeting 18th-21st December 2005, University of Oxford, Abstracts, p. 37-38.
J.-Y., P., F., S.Q., C.-W., D.J. & E.H. (2002).- Precambrian animal life: probable developmental and adult cnidarian forms from Southwest China.- Developmental Biology, Amsterdam, vol. 248, p. 182-196.
S., B.W. & M. (1990).- The early skeletal organism Cloudina: New occurrences from Oman and possibly China.- American Journal of Science, New Haven, vol. 290-A, p. 245-260.
I., M., S. & T. (2010).- A new species of Cloudina from the terminal Edicaran of Spain.- Precambrian Research, Amsterdam, vol. 176, p. 1-10.
W., Z. & W. (2003).- Diversification of skeletal microstructures of organisms through the interval from the latest Precambrian to the Early Cambrian.- Science in China, Beijing, (Series D), vol. 46, p. 977-985.
R., B. & J. (2000).- Organomineralization of cirratulid annelid tubes - fossil and recent examples.- Facies, Erlangen, vol. 42, p. 35-50.
C. & P. (1999).- Upper Vendian skeletal fauna of the Arroyo del Soldado Group, Uruguay.- Beringeria, Würzburg, vol. 23, p. 55-91.
J.G.B. (1972).- New shelly fossils from the Nama Group, South West Africa.- American Journal of Science, New Haven, vol. 272, p. 752-761.
M.F. (1976).- Early Phanerozoic annelid worms and their geological and biological significance.- Journal of the Geological Society, London, vol. 132, p. 259-275.
S.W.F. (1990).- Shell structure and distribution of Cloudina, a potential index fossil for the terminal Proterozoic.- American Journal of Science, New Haven, vol. 290-A, p. 261-294.
J.P., S.A., B.Z. & A.J. (1995).- Biostratigraphic and geochronologic constraints on early animal evolution.- Science, Washington, vol. 270, p. 598-604.
G. & H.D. (1985).- Die Cloudinidae n. fam., Kalk-Röhren aus dem Vendium und Unter-Kambrium.- Senckenbergiana Lethaia, Frankfurt, vol. 65, p. 413-431.
R.H. (1958).- Tube formation by Pomatoceros triqueter (Polychaeta).- Journal of the Marine Biological Association of the United Kingdom, Plymouth, vol. 37, p. 315-322.
H.A. ten & P. van den (1993).- A review of Recent and fossil serpulid 'reefs'; actuopalaeontology and 'Upper Malm' serpulid limestones in NW Germany.- Geologie en Mijnbouw, Utrecht, vol. 72, p. 23-67.
H.A. ten & E.K. (2009).- Taxonomy of Serpulidae (Annelida, Polychaeta): the state of affairs.- Zootaxa, Auckland, vol. 2036, p. 1-126.
H.A. ten & R.S. (1990).- A re-description of Ditrupa gracillima , 1878 (Polychaeta, Serpulidae) from the Indo-Pacific, with a discussion of the genus.- Records of the Australian Museum, Sydney, vol. 42, p. 101-118.
H., Z. & X. (2007).- The advent of mineralized skeletons in Neoproterozoic Metazoa— new fossil evidence from the Gaojiashan Fauna.- Geological Journal, Malden, vol. 42, p. 263-279.
H., Z., X., L. & S. (2005).- Skeletogenesis and asexual reproduction in the earliest biomineralizing animal Cloudina.- Geology, Boulder, vol. 33, p. 277-280.
H., B.R. & L. (2003).- Borings in Cloudina shells: Complex predator-prey dynamics in the terminal Neoproterozoic.- Palaios, Tulsa, vol. 18, p. 454-459.
M. (2004).- Serpulidae und Spirorbidae (Polychaeta sedentaria) aus Campan und Maastricht von Norddeutschland, den Niederlanden, Belgien und angrenzenden Gebieten.- Geologisches Jahrbuch, Stuttgart, vol. A157, p. 121-249.
P., S., C., F. & L. (2009).- Silicified tubular microfossils from the upper Doushantuo Formation (Ediacaran) in the Yangtze Gorges area, South China.- Journal of Paleontology, Tulsa, vol. 83, p. 630-633.
P., S., C., C., L. & F. (2008).- Systematic description and phylogenetic affinity of tubular microfossils from the Ediacaran Doushantuo Formation at Weng'an, South China.- Palaeontology, London, vol. 51, p. 339-366.
J.M. (1971).- Ultrastructural studies of the secretion of calcium carbonate by the serpulid polychaete worm, Pomatoceros caeruleus.- Zeitschrift für Zellforschung, New York, vol. 120, p. 160-186.
J.A. & K.R. (1975).- Calcium accumulation and secretion in the serpulid polychaete Spirorbis spirorbis at settlement.- Journal of the Marine Biological Association of the United Kingdom, Plymouth, vol. 55, p. 911-923.
J.S. (2011).- The coral Cothonion from the lower Cambrian of North Greenland.- Alcheringa, Abingdon, vol. 35, p. 405-411.
B. (2001).- Escape hatches for the clonal offspring of serpulid polychaetes.- Biological Bulletin, Woods Hole, vol. 200, p. 107-117.
R. (1996).- Micromorphology, microstructure and functional morphology of the Josephella marenzelleri (Polychaeta Serpulidae) tube. In: Cerchi A, (ed.), Autoecology of selected fossil organisms: achievements and problems.- Bolletino della Società Paleontologica Italiana, Special Volume, Modena, vol. 3, p. 205-211.
R. (2009).- Systematics and life habit in Serpula israelitica , 1977 (Polychaeta Serpulidae) from the Mediterranean with remarks on other soft-bottom serpulids.- Journal of Natural History, London, vol. 43, p. 2009-2025.
A.E., N., R.M.A., C.J. & G.C. (2010).- Insights into the composition, morphology, and formation of the calcareous shell of the serpulid Hydroides dianthus.- Journal of structural Biology, San Diego, vol. 169, p. 145-160.
P.D. (2008).- Seawater chemistry, biomineralization and the fossil record of calcareous organisms. In: H., S.F., N. & P. (ed.), Origin and evolution of natural diversity.- Proceedings of International Symposium "The origin and evolution of natural diversity", 1-5 October 2007, Sapporo, 21st Century COE for Neo-Science of Natural History, Hokkaido University, p. 21-29.
P.D. & O. (2006).- Convergent morphology in small spiral worm tubes ('Spirorbis') and its palaeoenvironmental implications.- Journal of the Geological Society, London, vol. 163, p. 225-228.
P.D., O. & M.A. (2010).- Evolution of biomineralization in "lophophorates".- Special Papers in Palaeontology, London, vol. 84, p. 317-333.
O. (2005).- A new cornulitid genus from the Silurian of Gotland, Sweden.- GFF, Stockholm, vol. 127, p. 205-210.
O. (2007).- Taxonomic implications and fossilization of tube ultrastructure of some Cenozoic serpulids (Annelida, Polychaeta) from Europe.- Neues Jahrbuch für Geologie und Paläontologie, Stuttgart, vol. 244, p. 115-128.
O. (2008).- Tube ultrastructure of the fossil genus Rotularia , 1827 (Polychaeta, Serpulidae).- Journal of Paleontology, Tulsa, vol. 82, p. 206-212.
O. (2009).- The ultrastructure of calcareous cirratulid (Polychaeta, Annelida) tubes.- Estonian Journal of Earth Sciences, Tallinn, vol. 58, p. 153-156.
O. (2010).- Adaptive strategies in the evolution of encrusting tentaculitoid tubeworms.- Palæogeography, Palæoclimatology, Palæoecology, Amsterdam, vol. 292, p. 211-221.
O. & H. (2008).- Tube structure and ultrastructure of serpulids from the Jurassic of France and Switzerland, its evolutionary implications.- Neues Jahrbuch für Geologie und Paläontologie, Stuttgart, vol. 250, p. 129-135.
O., H.A. ten & H. (2008a).- On the tube ultrastructure and origin of calcification in sabellids (Annelida, Polychaeta).- Palaeontology, London, vol. 51, p. 295-301.
O., H.A. ten, H. & K. (2008b).- Ultrastructure and mineral composition of serpulid tubes (Polychaeta, Annelida).- Zoological Journal of the Linnean Society, London, vol. 154, p. 633-650.
O., M. & K. (2008c).- Microscopic evidence of serpulid affinities of the problematic fossil tube "Serpula" etalensis from the Lower Jurassic of Germany.- Lethaia, Oslo, vol. 41, p. 417-421.
O., K. & H.A. ten (2009).- Tube ultrastructure of Pomatoceros americanus (Polychaeta, Serpulidae): implications for the tube formation of serpulids.- Estonian Journal of Earth Sciences, Tallinn, vol. 58, p. 148-152.
O. & H. (2009).- Calcareous tubeworms of the Phanerozoic.- Estonian Journal of Earth Sciences, Tallinn, vol. 58, p. 286-296.
O., H., H.A. ten & K. (2008d).- Unique Mg-calcite skeletal ultrastructure in the tube of the serpulid polychaete Ditrupa.- Neues Jahrbuch für Geologie und Paläontologie, Stuttgart, vol. 248, p. 79-89.
J., P. & D.E.G. (2008).- Machaeridians are Palaeozoic armoured annelids.- Nature, London, vol. 451, p. 185-188.
M.J. (1994).- Tube microstructure of recent and Jurassic serpulid polychaetes and the question of the Palaeozoic 'spirorbids'.- Acta Palaeontologica Polonica, Warsaw, vol. 39, p. 1-15.
L. (1986).- Preliminary study of the Gaojiashan biota from the latest Sinian in Ningqiang County, Shaanxi.- Bulletin of the Xi'an Institute of Geological Sciences, Xian, vol. 13, p. 67-88.
A.Yu., E., J.A., F. & A.B. (2011, online first).- New finds of skeletal fossils in the terminal Neoproterozoic of the Siberian Platform and Spain.- Acta Palaeontologica Polonica, Warsaw. DOI: 10.4202/app.2010.0074