Contents
[1. Introduction] [2. Geological background] [3. Locality and stratigraphy]
[4. Material and methods][5. Results]
[6. Discussion] [7. Conclusion] [Bibliographic references]
Department of Geology, University of Tartu, Ravila 14A, EE-50411 Tartu (Estonia)
Department of Geology, The College of Wooster, Wooster, 44691, Ohio (USA)
Manuscript online since October 2, 2012
[Editor and copy editor: Christian C. ]
A shallow shelf carbonate platform (pelletal limestone facies) stromatoporoid association from the late Sheinwoodian of Saaremaa (Baltica) contains a diverse assemblage of sclerobionts (both epi- and endobionts). The studied stromatoporoids vary from low domical to extended domical shapes. Cornulites sp. aff. C. stromatoporoides, Conchicolites sp., Anticalyptraea calyptrata, microconchids, tabulate (Aulopora sp., Catenipora sp. and favositids) and rugose corals, sheet-like trepostome bryozoans, and discoidal crinoid holdfasts encrust the stromatoporoids. The dominant sclerozoans were tabulate and rugose corals, which is significantly different from several analogous Silurian sclerobiont communities. There may have been taxonomic polarity between an upper surface and a cryptic sclerozoan community. Bioerosion occurs as macroborings in 45.5 % of studied (N=22) stromatoporoids. Endobionts were represented by Trypanites and Palaeosabella borings, as well as embedded symbiotic rugose corals and Cornulites stromatoporoides.
Sclerobionts; symbiosis; bioerosion; Silurian; Saaremaa; Baltica.
0. & M. A. (2012).- Encrustation and bioerosion on late Sheinwoodian (Wenlock, Silurian) stromatoporoids from Saaremaa, Estonia.- Carnets de Géologie [Notebooks on Geology], Brest, Article 2012/07 (CG2012_A07), p. 183-191.
Encroûtement et bioérosion de stromatoporoïdes d'âge Sheinwoodien supérieur (Wenlock, Silurien) de Saaremaa (Estonie).- Un peuplement de stromatoporoïdes de plate-forme carbonatée peu profonde (faciès calcaire péloïdal) d'âge Sheinwoodien supérieur (Wenlock inférieur) de Saaremaa (Estonie) recèle une association variée de sclérobiontes (épibiontes et endobiontes). Les stromatoporoïdes étudiés présentent différentes morphologies de dômes, basses à surélevées. Ces stromatoporoïdes sont encroûtés par Cornulites sp. aff. C. stromatoporoides, Conchicolites sp., Anticalyptraea calyptrata, des microconchidés, des tabulés (Aulopora sp., Catenipora sp. et des favositides), des tétracoralliaires, des bryozoaires trépostomes lamellaires et des crinoïdes dont subsistent les crampons discoïdes. Les sclérozoaires dominants sont des coraux (Tabulata et Rugosa), ce qui représente une différence significative par rapport aux nombreuses communautés analogues de sclérobiontes siluriens. Il y a pu avoir une polarité taxonomique entre une surface supérieure et une communauté cryptique de sclérozoaires. Sur les stromatoporoïdés étudiés (N=22), 45.5 % des cas de bioérosion correspondent à des perforations de grande taille. Les endobiontes sont représentés par des perforations de types Trypanites et Palaeosabella, ainsi que par des formes "enchâssées", tétracoralliaires (Rugosa) et Cornulites stromatoporoides.
Sclérobiontes ; symbiose ; bioérosion ; Silurien ; Saaremaa ; Baltica ; Fenno-sarmatia.
Marine invertebrate communities diversified rapidly during the Great Ordovician Biodiversification Event (GOBE; see, for review, et al., 2009). There was also a dramatic increase in encrustation and bioerosion on hard substrates at this time ( & , 2003). The diversification of bioeroders has been called the Ordovician Bioerosion Revolution ( & , 2006). The Silurian was a time of further development of hard substrate communities after the GOBE. In general, Silurian hard substrate communities are similar to those of the Ordovician, being primarily dominated by bryozoans and echinoderms ( & , 2003). Encrusting faunas of the Silurian stromatoporoids have been studied in detail by several authors (, 1980; & , 1988; , 2000). Encrustation patterns and bioerosion is relatively well known for western Baltica, especially for Gotland, Sweden (, 1980; , 1984; et al., 2008). However, little published data on sclerobionts are available from the Silurian of eastern Baltica (, 1970; & , 2010a, 2010b).
The aims of this paper are: 1) to describe a hard substrate association from the late Sheinwoodian of East Baltic for the first time; 2) to discern whether this association is typical for the Silurian of Baltica and beyond; 3) to discover whether hard substrate cryptic niches were occupied; and 4) to determine whether the bioerosion was similar to that observed in the Silurian of North America and western Baltica (especially Gotland).
During the Silurian the Baltica continent was located in equatorial latitudes drifting northwards ( et al., 2004). The pericontinental Baltica paleobasin in modern Estonia was characterized by a wide range of tropical environments and diverse biotas ( et al., 2008). In the outcrop area, including Saaremaa Island, the Silurian succession is represented by shallow shelf carbonate rocks rich in shelly faunas. The Silurian exposures on Saaremaa, which are the best in Estonia, are mostly represented by coastal cliffs.
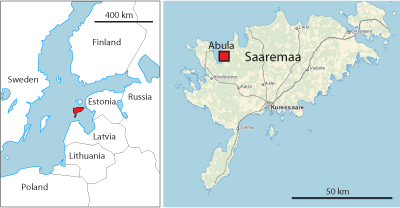
The Abula cliff is situated on the eastern coast of Tagalaht Bay, 3 km north of the Mustjala-Veere road (58°27'12" N, 22°06'51" E) (Fig. 1 ![]() ). The cliff exposes the topmost Vilsandi Beds (lagoonal dolomitic marlstones) and the basal part of the Maasi Beds ( et al.,
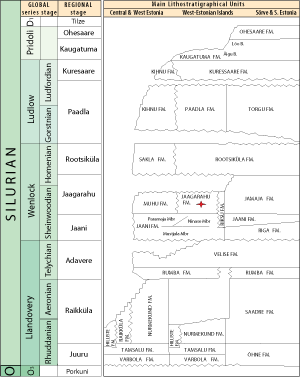
2008). The Maasi Beds form the middle part of the Jaagarahu Formation (Figs. 2
). The cliff exposes the topmost Vilsandi Beds (lagoonal dolomitic marlstones) and the basal part of the Maasi Beds ( et al.,
2008). The Maasi Beds form the middle part of the Jaagarahu Formation (Figs. 2 ![]() - 3
- 3 ![]() ). The studied stromatoporoids were collected from the pelletal limestone layers of the upper part of the section (the Maasi Beds). The stromatoporoid-rich layer is of normal marine origin. It is presumably deposited on a very shallow shelf in waves activity zone, what is indicated by occurrence of rare overturned stromatoporoids.
). The studied stromatoporoids were collected from the pelletal limestone layers of the upper part of the section (the Maasi Beds). The stromatoporoid-rich layer is of normal marine origin. It is presumably deposited on a very shallow shelf in waves activity zone, what is indicated by occurrence of rare overturned stromatoporoids.
Click on thumbnail to enlarge the image.
Figure 1: Location of Abula cliff (Maasi Beds, Jaagarahu Formation, late Sheinwoodian) on Saaremaa Island, Estonia. The map of Saaremaa is provided by Estonian Land Board.
Click on thumbnail to enlarge the image.
Figure 2: Stratigraphy of the Silurian of Estonia (after et al., 2008). Location of Maasi Beds with stromatoporoids marked with red star.

Click on thumbnail to enlarge the image.
Figure 3: Abula cliff, Maasi Beds, Jaagarahu Formation. Stromatoporoid-rich horizon is indicated by yellow lines.
Twenty-two stromatoporoids were collected from the pelletal limestone layers (Maasi Beds) of Abula cliff. Sample orientations were marked during collecting. The specimens were manually cleaned with water and a soft brush. Trypanites-Palaeosabella borings and endobiotic rugosans were counted in a 4 cm2 grid. The surface areas of encrusting fossils were estimated using a millimeter grid. A centimeter grid was used to calculate the total surface area of the stromatoporoids. Upper surface and cryptic encrustations were measured separately.
Our classification of stromatoporoid shapes follows (1998). Three morphological categories of stromatoporoids were identified: 1) low domical, 2) high domical, and 3) extended domical. The preservation of our material is variable, and it is possible that some stromatoporoids may have lost some part of their original encrusters while broken free from the limestone matrix. Thus, encrustation densities on stromatoporoids reported here are minimums.
Endobiotic cornulitids from the same association were described by and (2010c) and were not counted in this study as their abundance was determined by a different method (breaking the stromatoporoids into pieces by hammer).
Figured specimens are deposited at the Geological Museum, Museum of Natural History, University of Tartu (TUG).
| Taxon | Upper surface Number of specimens | Cryptic surface Number of specimens | Total number of specimens | Number of stromatoporoids with particular encruster |
| Cornulites aff. stromatoporoides | 8 (13.3%) | - | 8 (10.7%) |
2 |
| Anticalyptraea calyptrata | - | 2 (13.3%) | 2 (2.7%) |
1 |
| Conchicolites sp. | - | 1 (6.7%) | 1 (1.3%) |
1 |
| Microconchids | 15 (25%) | - | 15 (20%) |
1 |
| Sheet-like trepostome bryozoans | 3 (5%) | 4 (26.7%) | 7 (9.3%) |
5 |
| Rugosans | 17 (28.3%) | - | 17 (22.7%) |
6 |
| Auloporids | 5 (8.3%) | 7 (46.6%) | 12 (16%) |
5 |
| Favositids | 6 (10%) | 1 (6.7%) | 7 (9.3%) |
5 |
| Catenipora sp. | 2 (3.3%) | - | 2 (2.7%) |
2 |
| Crinoid holdfast (discoidal) | 4 (6.7%) | - | 4 (5.3%) |
3 |
|
Total |
60 |
15 |
75 |
Table 1: Faunal composition of the stromatoporoid sclerobiont association.
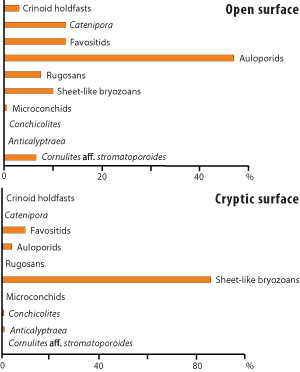
Encrusting tentaculitoid tubeworms (Cornulites sp. aff. C. stromatoporoides, Conchicolites sp., microconchids), tabulate corals (Aulopora sp., Catenipora sp., favositids), rugose corals, sheet-like trepostome bryozoans, and discoidal crinoid holdfasts were found encrusting the stromatoporoids. C. aff. stromatoporoides, microconchids, Catenipora sp., rugosans, and crinoid holdfasts were attached only to the upper surface of stromatoporoids, while Anticalyptraea calyptrata and Conchicolites sp. were found only on cryptic surfaces (those other than upward-facing) (Table 1; Fig. 4 ![]() ). Corals are numerically the most important encrusters (50.7 %, N=38) (Fig. 5
). Corals are numerically the most important encrusters (50.7 %, N=38) (Fig. 5 ![]() ), followed by tentaculitoid tubeworms (34.7 %, N=26) and bryozoans (9.3 %, N=7) (Fig. 6
), followed by tentaculitoid tubeworms (34.7 %, N=26) and bryozoans (9.3 %, N=7) (Fig. 6 ![]() ). Numerically least important are crinoids (5.3 %, N=4) (Fig. 7
). Numerically least important are crinoids (5.3 %, N=4) (Fig. 7 ![]() ). There is a difference in taxonomic composition between the upper surface community and the cryptic community.
). There is a difference in taxonomic composition between the upper surface community and the cryptic community.

Click on thumbnail to enlarge the image.
Figure 4: Diagrams showing abundance of sclerobionts on Abula cliff stromatoporoids.

Click on thumbnail to enlarge the image.
Figure 5: A favositid coral colony cemented to the cryptic surface of a stromatoporoid. Abula cliff, Saaremaa (Estonia), Maasi Beds, Jaagarahu Formation, late Sheinwoodian. TUG 1627-1.

Click on thumbnail to enlarge the image.
Figure 6: Bryozoans on the cryptic surface of a high domical stromatoporoid. Abula cliff, Saaremaa (Estonia), Maasi Beds, Jaagarahu Formation, late Sheinwoodian. TUG 1627-2.
The stromatoporoids in this study are sparsely covered by skeletal sclerobionts. Both upper (2.5 % encrusted) and cryptic surfaces (3.5 % encrusted) are similar in this respect (Table 2; Fig. 8 ![]() ). Colonial encrusters have the largest area of skeletal cover. In the upper surface community, tabulate corals dominate by area of skeletal cover (72.2 %), followed by bryozoans (10.1 %). The least important in the upper surface community by area of skeletal cover are crinoids (3.0 %). In the cryptic community, bryozoans have the largest area of skeletal cover (85.8 %) followed by tabulate corals (13 %) (Table 2). Skeletal overgrowth was not observed between the encrusters in either the open or cryptic communities.
). Colonial encrusters have the largest area of skeletal cover. In the upper surface community, tabulate corals dominate by area of skeletal cover (72.2 %), followed by bryozoans (10.1 %). The least important in the upper surface community by area of skeletal cover are crinoids (3.0 %). In the cryptic community, bryozoans have the largest area of skeletal cover (85.8 %) followed by tabulate corals (13 %) (Table 2). Skeletal overgrowth was not observed between the encrusters in either the open or cryptic communities.
| Upper surface area studied 2588 cm2 | Cryptic surface area studied 373 cm2 | ||||
| Taxa | Area covered cm2 | % of skeletal cover | Area covered cm2 | % of skeletal cover | |
| Cornulites aff. stromatoporoides | 4.2 | 6.5 | |||
| Anticalyptraea calyptrata | 0.1 | 0.8 | |||
| Conchicolites sp. | 0.05 | 0.4 | |||
| Microconchids | 0.3 | 0.5 | |||
| Sheet-like trepostome bryozoans | 6.5 | 10.1 | 11.2 | 85.8 | |
| Rugosans | 4.9 | 7.6 | |||
| Auloporids | 30.2 | 47 | 0.5 | 3.8 | |
| Favositids | 8.1 | 12.6 | 1.2 | 9.2 | |
| Catenipora sp. | 8.1 | 12.6 | |||
| Crinoid holdfast (discoidal) | 1.9 | 3.0 | |||
|
Total area encrusted |
64.2 cm2 (2.5 %) |
13.05 cm2 (3.5%) |
|||
Table 2: Skeletal cover of upper and cryptic surfaces of the stromatoporoids.
Click on thumbnail to enlarge the image.
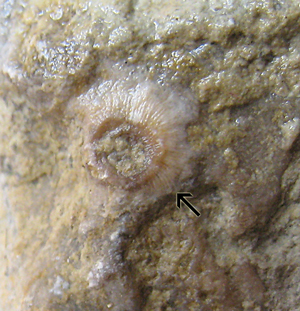
Figure 7: Discoidal crinoid holdfast on the upper surface of an extended domical stromatoporoid. Abula cliff, Saaremaa (Estonia), Maasi Beds, Jaagarahu Formation, late Sheinwoodian. Diameter of the disc 9 mm. TUG 1627-3.
Click on thumbnail to enlarge the image.
Figure 8: Percent of skeletal cover of upper and cryptic surfaces of the stromatoporoids by sclerobiont taxa.
Endobiotic rugosan symbionts were found in two stromatoporoids (9.1 %, N=22) (Figs. 9 ![]() - 10
- 10 ![]() ). Both stromatoporoids with endobiotic rugosans have a low domical shape. There are 10 to 20 endobiotic rugosans per 4 cm2, counting more than 100 rugosans per stromatoporoid. The apertures of the rugosans are 0.5 to 3.0 mm wide and are usually situated at the level of the surrounding stromatoporoid surface. A few rugosans show slightly elevated apertures (< 0.5 mm). The endobiotic rugosans are spread all over the stromatoporoid upper surface without any preference for certain regions.
). Both stromatoporoids with endobiotic rugosans have a low domical shape. There are 10 to 20 endobiotic rugosans per 4 cm2, counting more than 100 rugosans per stromatoporoid. The apertures of the rugosans are 0.5 to 3.0 mm wide and are usually situated at the level of the surrounding stromatoporoid surface. A few rugosans show slightly elevated apertures (< 0.5 mm). The endobiotic rugosans are spread all over the stromatoporoid upper surface without any preference for certain regions.

Click on thumbnail to enlarge the image.
Figure 9: Symbiotic rugosan corals in a stromatoporoid in longitudinal section. Abula cliff, Saaremaa (Estonia), Maasi Beds, Jaagarahu Formation, late Sheinwoodian. TUG 1627-4.
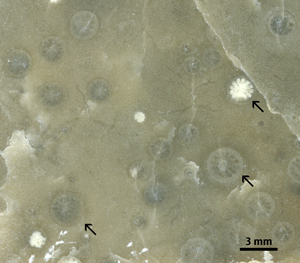
Click on thumbnail to enlarge the image.
Figure 10: Symbiotic rugosan corals in a stromatoporoid in cross section. Abula cliff, Saaremaa (Estonia), Maasi Beds, Jaagarahu Formation, late Sheinwoodian.
Ten stromatoporoids of 22 studied (45.5 %) were bioeroded, mostly by Trypanites (Fig. 11 ![]() ). Among ten exposed borings, one resembled Palaeosabella. Maximum boring density was eight borings per 4 cm2, but average density was one to five for the same area. Palaeosabella differs from cylindrical Trypanites by its flared distal end. Palaeosabella-Trypanites range from 0.30 to 4.00 mm in diameter and extend to a maximum depth of 1.0 cm below the substrate surface. Distribution of the borings is patchy on the stromatoporoid upper surfaces. In high domical and extended domical stromatoporoids, borings are often more numerous in the most elevated part of the substrate.
). Among ten exposed borings, one resembled Palaeosabella. Maximum boring density was eight borings per 4 cm2, but average density was one to five for the same area. Palaeosabella differs from cylindrical Trypanites by its flared distal end. Palaeosabella-Trypanites range from 0.30 to 4.00 mm in diameter and extend to a maximum depth of 1.0 cm below the substrate surface. Distribution of the borings is patchy on the stromatoporoid upper surfaces. In high domical and extended domical stromatoporoids, borings are often more numerous in the most elevated part of the substrate.

Click on thumbnail to enlarge the image.
Figure 11: Trypanites borings in the upper surface of a high domical stromatoporoid. Abula cliff, Saaremaa (Estonia), Maasi Beds, Jaagarahu Formation, late Sheinwoodian. TUG 1627-5.
Both bioerosional trace fossils, Trypanites and Palaeosabella, had a global distribution in the Silurian ( & , 2003). Among the Abula cliff stromatoporoids, Trypanites borings are most prominent, which is similar to the situation in the Silurian of Gotland (western Baltica) (, 1984) and Anticosti (North America) ( et al., 2004). et al. (2004) found for the Silurian of Anticosti Island that samples from the reef are bored least frequently (28 %). Among the sandy off-reef samples, they found 41 % bored substrates, and 50 % of muddy facies samples were bored. The boring frequency among Abula cliff stromatoporoids (45.5 %) is similar to both off-reef samples described by et al. (2004). However, the Abula cliff pelletal limestones can best be compared to the sandy off-reef facies by et al. (2004). Similarity in the boring frequency of large skeletal substrates between North America and Baltica in the early Silurian is not surprising considering their similar paleoclimates and paleogeographic proximity. The low bioerosion density for the Silurian ( et al., 2004) presumably indicates a relatively short exposure time of the Abula stromatoporoids to borers, or a relatively small number of borers in the community, or extensive occupation of the stromatoporoid surfaces by soft-bodied encrusters. Occurrence of more numerous borings in the most elevated parts of the substrate in high domical and extended domical stromatoporoids has several known analogues. The organisms that produced Trypanites borings preferentially drilled the high points on hard substrates in the Ordovician and Silurian, probably for better filter-feeding opportunities ( & , 1978; & , 1984; , 1984; & , 1992).
The taxonomic composition of encrusters recorded on the Abula stromatoporoids is characteristic of the Silurian (Table 1; Fig. 4 ![]() ). However, there are some peculiarities among the Abula stromatoporoid encrusters considering the numerical abundance and relative area covered by skeletons. In general, typical Silurian encrusting faunas were similarly to those of the Ordovician dominated by bryozoans and echinoderms ( & ,
2003). In contrast, the Abula stromatoporoids show a very high number of corals (50.7%, N=38) and tentaculitoid tubeworms (34.7 %, N=26) in the association. Corals also have the largest area of skeletal cover in the upper surface community. Though numerically important, tentaculitoid tubeworms covered only small areas on the stromatoporoids as compared to tabulate corals and bryozoans. Echinoderms are both numerically and by area of skeletal cover the least important encrusters on the Abula stromatoporoids (Tables 1
- 2; Figs. 4
). However, there are some peculiarities among the Abula stromatoporoid encrusters considering the numerical abundance and relative area covered by skeletons. In general, typical Silurian encrusting faunas were similarly to those of the Ordovician dominated by bryozoans and echinoderms ( & ,
2003). In contrast, the Abula stromatoporoids show a very high number of corals (50.7%, N=38) and tentaculitoid tubeworms (34.7 %, N=26) in the association. Corals also have the largest area of skeletal cover in the upper surface community. Though numerically important, tentaculitoid tubeworms covered only small areas on the stromatoporoids as compared to tabulate corals and bryozoans. Echinoderms are both numerically and by area of skeletal cover the least important encrusters on the Abula stromatoporoids (Tables 1
- 2; Figs. 4 ![]() & 8
& 8 ![]() ). Microconchids are common encrusters in the Silurian. The presence of microconchids in large numbers only on a single stromatoporoid specimen could either be explained by an original patchy distribution (gregariousness) or by weak preservation potential compared to other encrusters with more massive skeletons. The dominance of corals over the bryozoans make the Abula stromatoporoid epibiont fauna different from the reef stromatoporoid fauna of the Ludlow of Gotland ( & ,
1988) where bryozoans dominate the upper surface community.
). Microconchids are common encrusters in the Silurian. The presence of microconchids in large numbers only on a single stromatoporoid specimen could either be explained by an original patchy distribution (gregariousness) or by weak preservation potential compared to other encrusters with more massive skeletons. The dominance of corals over the bryozoans make the Abula stromatoporoid epibiont fauna different from the reef stromatoporoid fauna of the Ludlow of Gotland ( & ,
1988) where bryozoans dominate the upper surface community.
There is a likely taxonomic polarity between the upper surface and cryptic communities in the Abula stromatoporoids. Most notable is a lack of rugose corals on the cryptic surfaces (Table 1; Fig. 4 ![]() ). Similarly, a taxonomic polarity of epibionts has been reported from the reef stromatoporoids from the Ludlow of Gotland ( & ,
1988). Anticalyptraea has previously been reported from cryptic surfaces under the late Silurian hardground from Ohesaare, Saaremaa ( & ,
2010a). Cryptic surfaces in the Silurian were similar to the Ordovician, being mostly populated by the bryozoans, and cornulitids may also have been locally important (,
1980; & ,
2003). Considering the small cryptic area studied, it is impossible to draw firm conclusions for the Abula stromatoporoids, but bryozoans were also found to be dominant encrusters by area of skeletal cover (85.8%) (Table 2; Fig. 8
). Similarly, a taxonomic polarity of epibionts has been reported from the reef stromatoporoids from the Ludlow of Gotland ( & ,
1988). Anticalyptraea has previously been reported from cryptic surfaces under the late Silurian hardground from Ohesaare, Saaremaa ( & ,
2010a). Cryptic surfaces in the Silurian were similar to the Ordovician, being mostly populated by the bryozoans, and cornulitids may also have been locally important (,
1980; & ,
2003). Considering the small cryptic area studied, it is impossible to draw firm conclusions for the Abula stromatoporoids, but bryozoans were also found to be dominant encrusters by area of skeletal cover (85.8%) (Table 2; Fig. 8 ![]() ). In contrast to the Ohesaare hardground (Pridoli) and reef stromatoporoid from Gotland ( & ,
1988), no clear difference in skeletal cover was observed in the Abula stromatoporoids between cryptic and upper surface communities.
). In contrast to the Ohesaare hardground (Pridoli) and reef stromatoporoid from Gotland ( & ,
1988), no clear difference in skeletal cover was observed in the Abula stromatoporoids between cryptic and upper surface communities.
et al. (2002) found that modern bioerosion is higher at more productive sites in the ocean. They also found that more productive sites have higher encrustation rates. The Abula stromatoporoids have a relatively low encrustation density ( & , 1988; , 2000) combined with low bioerosion density for the Silurian ( et al., 2004). Thus, if these relations were valid in the late Silurian, the relatively low skeletal coverage of the stromatoporoids as compared to the other Ordovician to Devonian analogues ( & , 1978) may be explained by low productivity (low nutrient levels) in the sea water ( et al., 2002) or by a large number of soft-bodied organisms in the community that were not preserved.
& (2010c) described a cornulitid (Cornulites stromatoporoides) and stromatoporoid association from the Abula cliff assemblages. They found symbiotic cornulitid endobionts in 77 % of studied stromatoporoids. In maximum there were three cornulitid specimens counted per stromatoporoid host. The symbiotic rugosan endobionts here are more rare compared to the cornulitids, occurring only in 9 % (N=22) of studied stromatoporoids. However, their total number in the population is not insignificant as there may be about a hundred rugosans per infested stromatoporoid host. Similar stromatoporoid-rugose associations are known from the Silurian of Gotland (, 1969, 1970; , 1981, 1987) and the Devonian of Spain ( & , 1985). These associations are interpreted as commensal relationships by these authors. Similarly to endobiotic rugosan symbionts from the Silurian of Gotland (, 1987), the Abula endobiotic rugosans have small apertural diameters (0.5-3.0 mm). Further studies should show whether this rugosan-coral symbiotic relationship occurred only between certain taxa. The presence of both symbiotic cornulitid and rugosan endobionts indicates that symbiotic interactions were important among the Abula stromatoporoids. However, it is difficult to identify with fossil material whether this syn vivo association of stromatoporoids and rugosans was parasitic or mutualistic (, 2011).
1) The Sheinwoodian Abula sclerobiont assemblage differs from the analogous western Baltic ( & , 1988) and North American equivalents (, 2000) by dominance of corals in the assemblage.
2) Bioerosion on the Abula stromatoporoids was similar to the Early Silurian of North America (, 2004) and Gotland (, 1984), but the density of Trypanites-Palaeosabella boring was low in Abula as compared to the maxima in North America.
3) The Abula cliff stromatoporoids are rich in endobiotic symbionts, including both cornulitids and rugosans.
O.V. is indebted to the Grant (Paleontological Society), Estonian Science Foundation grant ETF9064 and the target-financed project (from the Estonian Ministry of Education and Science) SF0180051s08 (Ordovician and Silurian climate changes, as documented from the biotic changes and depositional environments in the Baltoscandian Palaeobasin) for financial support. M.W. thanks "The College of Wooster" for supporting his work in Estonia through the Luce and Wengerd Funds. We are grateful to Tomasz , Michał and Mikołaj for constructive reviews.
L., M., A. & A. (2008).- A giant boring in a Silurian stromatoporoid analyzed by computer tomography.- Acta Palaeontologica Polonica, Warsaw, vol. 53, p. 149–160.
C.E. & M.E. (1984).- Morphology, faunas and genesis of Ordovician hardgrounds from southern Ontario, Canada.- Palaeogeography, Palaeoclimatology, Palaeoecology, Amsterdam, vol. 46, p. 233–290.
C.E. & W.D. (1978).- Preservation and paleoecology of a Middle Ordovician hardground community.- Paleobiology, Tulsa, vol. 4, p. 329–348.
O., L., P. & T. (eds.) (2008).- The Seventh Baltic Stratigraphical Conference.- Abstracts and Field Guide, Geological Society of Estonia, Tallinn, 46 p.
D. (1970).- The Silurian of Estonia.- Valgus, Tallinn, 343 p. [in Russian]
S. (1980).- Cavities and cryptic faunas beneath non-reef stromatoporoids.- Lethaia, Oslo, vol. 13, p. 327-338.
S. (1981).- Stromatoporoid growth form and taxonomy in a Silurian biostrome.- Journal of Paleontology, Tulsa, vol. 55, p. 1284-1295.
S. (1987).- Stromatoporoid–coral intergrowths in a Silurian biostrome.- Lethaia, Oslo, vol. 20, p. 371– 380.
S. (1998).- The applications of stromatoporoid palaeobiology in palaeoenvironmental analysis.- Palaeontology, London, vol. 41, p. 509–544.
J.G. (2000).- Quantitative analysis of epizoans on Silurian stromatoporoids within the Brassfield Formation.- Journal of Paleontology, Tulsa, vol. 74, p. 394–403.
H.L., E. & M.J. (2002).- Mollusc shell encrustation and bioerosion rates in a modern epeiric sea: taphonomy experiments in the Java Sea, Indonesia.- Palaios, Lawrence, vol. 17, p. 171–191.
M.J., R.A. & P.M. (2004).- The Silurian Period. In: F.M. J.G. & A.G. (eds). A Geologic Time Scale 2004.- Cambridge University Press, p. 188-201.
K. (1969).- Stromatoporoids from the Silurian of Gotland, Part I.- Stockholm Contributions in Geology, Stockholm, vol. 19, p. 1-100.
K. (1970).- Stromatoporoids from the Silurian of Gotland, Part II.- Stockholm Contributions in Geology, Stockholm, vol. 22, p. 1-152.
E.W. (1984).- The boring of Silurian stromatoporoids-towards an understanding of larval behavior in the Trypanites organism.- Palæogeography, Palæoclimatology, Palæoecology, Amsterdam, vol. 48, p. 229–243.
M.T. & W.D. (1988).- Microhabitat analyses of Silurian stromatoporoids as substrata for epibionts.- Palaios, Lawrence, vol. 3, p. 391–403.
T., D.A.T., A., A.W. & P.M. (2009).- Understanding the Great Ordovician Biodiversification Event (GOBE): influences of paleogeography, paleoclimate, or paleoecology.- GSA Today, Boulder, vol. 19, p. 4-10.
F & I. (1985).- Estudio de una asociación coral rugoso-estromatopórido en el arrecife de Arnao (Fm Moniello, Asturias, NO de España).- Trabajos de Geología, Oviedo, vol. 15, p. 203-209.
L., P. & E. (2004).- Environmental and substrate control on Paleozoic bioerosion in corals and stromatoporoids, Anticosti Island, eastern Canada.- Palaios, Lawrence, vol. 19, p. 292–306.
P.D. & M.A. (2003).- Palaeoecology and evolution of marine hard substrate communities.- Earth Science Reviews, Amsterdam, vol. 62, p. 1–103.
O. & M.A. (2010a).- Microconchid-dominated hardground association from the late Pridoli (Silurian) of Saaremaa, Estonia.- Palaeontologia Electronica, Calvert, 13.2.9A, http://palaeo-electronica.org/2010_2/212/
O. & M.A. (2010b).- Occurrence of giant borings of Osprioneides kampto in the lower Silurian (Sheinwoodian) stromatoporoids of Saaremaa, Estonia.- Ichnos, Philadelphia, vol. 17, p. 166-171.
O. & M.A. (2010c).- Endosymbiotic Cornulites in the Sheinwoodian (Early Silurian) stromatoporoids of Saaremaa, Estonia.- Neues Jahrbuch für Geologie und Paläontologie, Abhandlungen, Stuttgart, vol. 257, p. 13–22.
M.A. & T.J. (1992).- Hardgrounds and hardground faunas.- Institute of Earth Studies Publications, University of Wales, Aberystwyth, vol. 9, 131 p.
M.A. & T.J. (2006).- Patterns and processes in the Ordovician Bioerosion Revolution.- Ichnos, Philadelphia, vol. 13, p. 109–112.
M.K. (2011).- Is absence of proof a proof of absence? Comments on commensalism.- Palæogeography, Palæoclimatology, Palæoecology, Amsterdam, vol. 302, p. 484–488.